
Nitinol Film:
From Research to Clinical Trial
• Started PhD research
• Aim: Fabricate Nitinol Stents with thin film technology
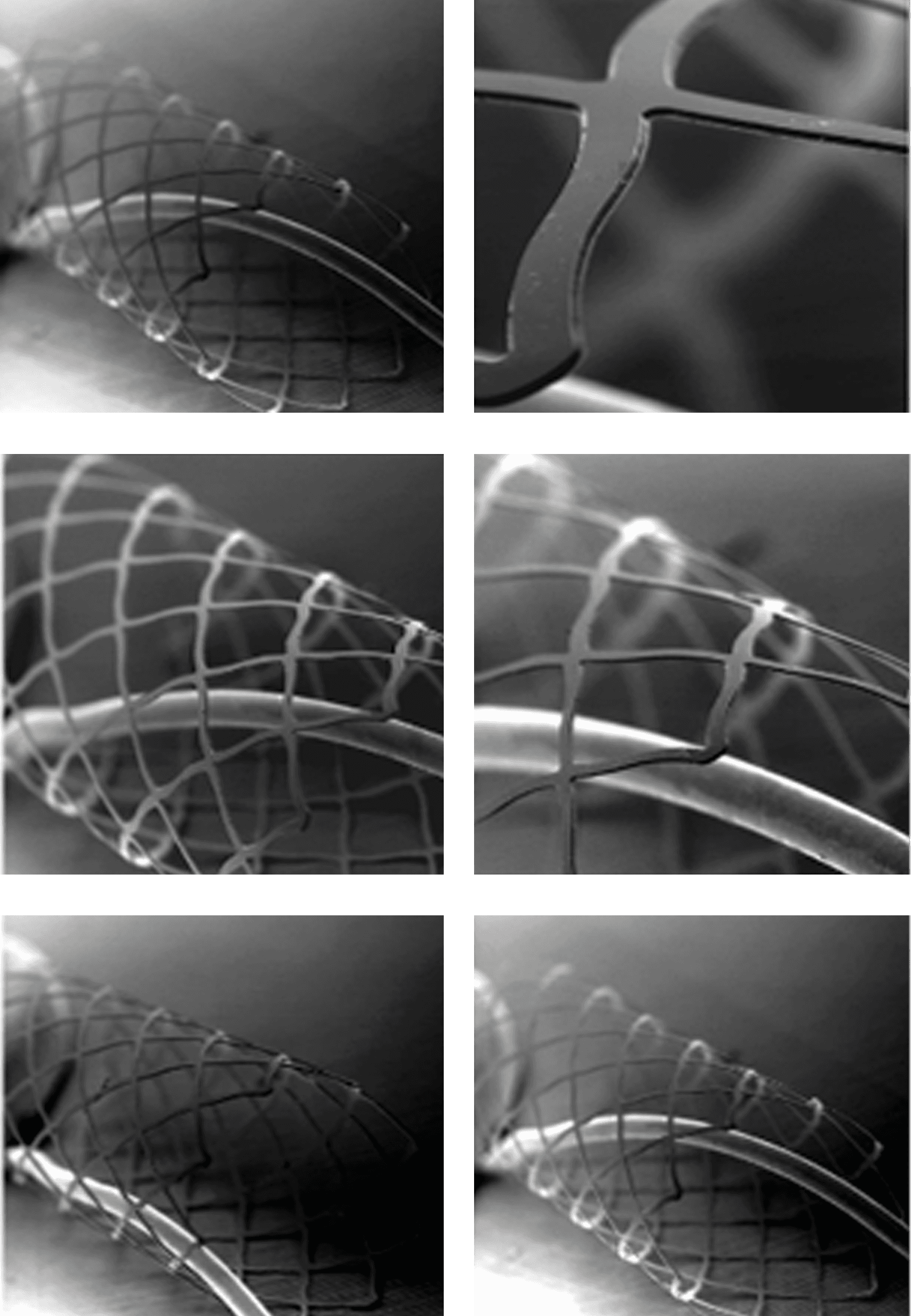
• PhD completed with summa cum laude distinction
• 1st Prize, WT.SH Idea Competition (Schleswig-Holstein)
• Inspired to explore founding a company based on research outcomes
• Core technology continued to mature toward application readiness

• Company Foundation
• Continued technology development
• First customer projects acquired
• Operated without external funding
• Published 20+ peer-reviewed scientific papers
• 12 registered patents
• Ongoing technology development and maturation
• Awarded by Werner Petersen Stiftung for innovation
• Winner of Medtec Europe Start-up Academy Award
• Progress in scalability, process development, and SOPs
• First Pilot serial production
• Team grew to 5 employees
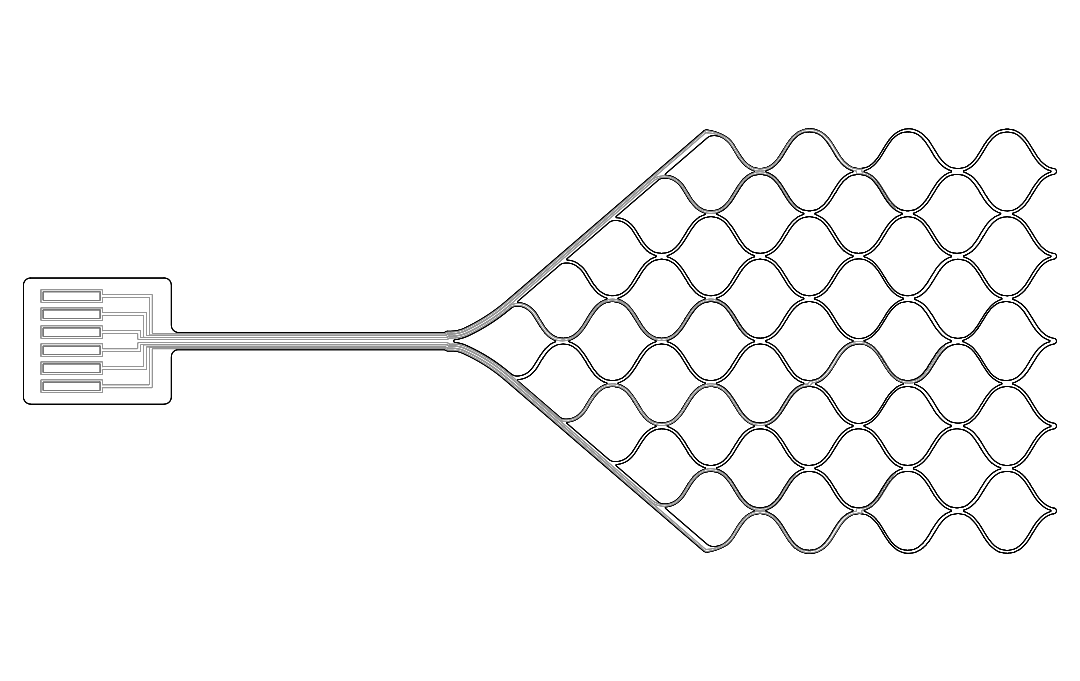
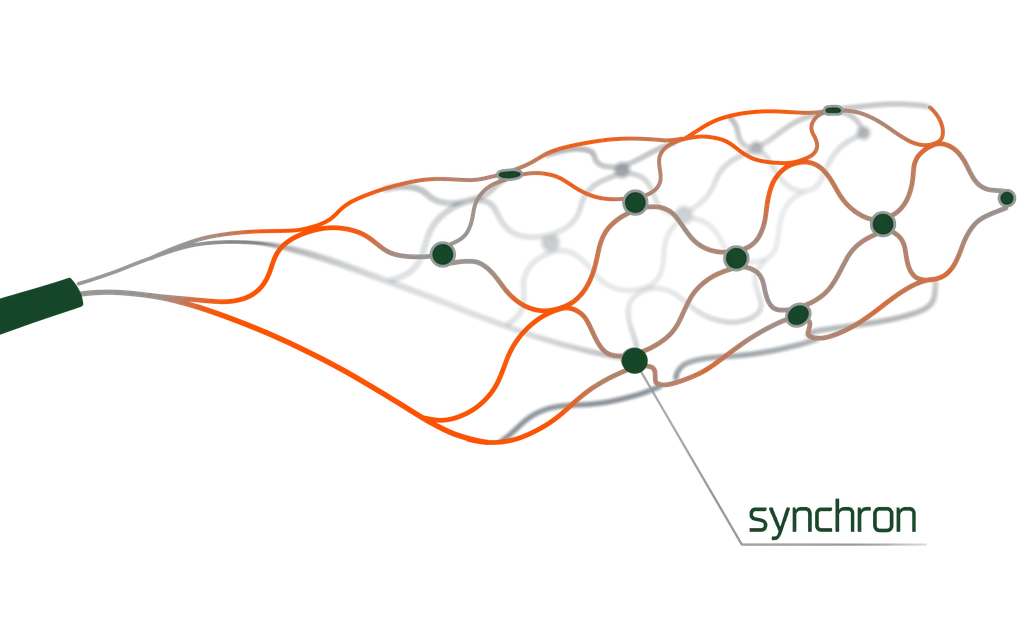
• FDA Breakthrough Device designation (bioelectronic application)
• First-in-human trial initiated (Pilot Study - Customer)
• Full QMS implementation (ISO 13485 compliant)
• Expansion of fabrication capabilities
• Broadened technology platform
• Achieved first market entry
• Team grew to 20 employees
• Closed funding round to support scale-up
• Initiated planning of new cleanroom facility
• Prepared ISO 13485 certification for new site
• Supported clinical trials for 3 clients
• Team expanded to 28 employees
• Completion and operational launch of new ACQUANDAS facility
• ISO 13485 certification (2027)

Our Leadership

Founder & CEO
Dr.-Ing. Rodrigo Lima de Miranda

Shareholder
Prof. Dr.- Ing. Eckhard Quandt

Shareholder & Managing Director of ACANDIS GmbH




















